Atomic structure powerpoints8/13/2023  
Determine chemical properties and the elements ability to form bonds.Electrons in the outermost energy level.p orbital: can hold 6 electrons (dumbbell shaped).s orbital: lowest energy level can hold 2 electrons (sphere shaped).orbits you can predict the location of an orbit, you cannot predict the orbital location. Modern theory has electrons in orbitals vs.Outer Electrons have more energy because they take the same time to orbit the nucleus but have to go a greater distance so the are faster.Atoms are really, really tiny: 109 atoms lined up = 1 cm.Electron cloud- region where you might find an electron.Must gain energy to move to higher orbit, lose energy to move to lower orbit.Each electron has a certain energy, determined by its path around nucleus.Slide 4 / 100 Matter You will recall that we define matter as anything that has mass and takes up space. The type of matter that is changing and what types of changes it undergoes determines the field of chemistry that is being studied. Bohr’s Model: Theorized that electrons were in set paths around nucleus like planets around sun (orbits). Atomic Structure Slide 3 / 100 Chemistry Chemistry is the study of matter and the changes it undergoes.Have an equal number of protons and electrons.Properties of Subatomic Particles Guaranteed To Be On Test Analogy: if the nucleus of an atom were the size of a marble, the atom would be the size of a football stadium.These are almost identical in size and mass, but different in charges.Nucleus: small, dense center which holds a positive charge. Atomic Structure Unit Plan (122 Favorites) LESSON PLAN in Atomic Spectra, Model of the Atom, Isotopes, Atomic Theory, Subatomic Particles, Emission Spectrum, Electrons, Orbitals, Ions, Unit Plans.Chemical reactions are rearrangement of atoms. 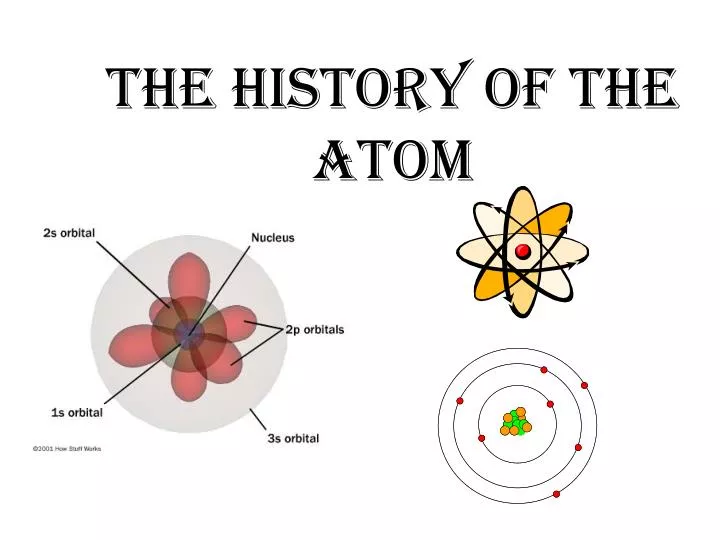
Each compound has a specific number and kinds of atom. Ability to add personal text in allocated text boxes or. Compounds are formed when atoms combine. Presentation for middle school to high school advanced as introduction to atomic structure.Atoms of different elements are different. Considered the foundation of modern Atomic Theory.Stated that atoms of the same element were all alike, and could join to form compounds.Proposed that atoms could not be divided.The movements of these atoms caused changes in matter.Suggested that the universe was made of invisible units named atoms. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
